What Is The Chemical Makeup Of Barbiturates
Barbiturates and Benzodiazepines
- Page ID
- 413
Hypnotic and sedative drugs are non-selective, full general depressants of the central nervous organisation. If the dose is relatively low, a sedative action results in a reduction in restlessness and emotional tension. A larger dose of the same drug produces a hypnotic sleep inducing issue. As the dosage is increased farther, the outcome is anesthesia or death if the dosage is sufficiently high.
Introduction
The barbiturates once enjoyed a long flow of extensive apply equally allaying-hypnotic drugs; however, except for a few specialized uses, they have been largely replaced past the much safer benzodiazepines. Barbiturates are central nervous organization depressants and are similar, in many ways, to the depressant effects of alcohol. To date, there are about 2,500 derivatives of barbituric acrid of which only fifteen are used medically. The first barbiturate was synthesized from barbituric acid in 1864. The original use of barbiturates was to replace drugs such as opiates, bromides, and alcohol to induce sleep.
The hyponotic and sedative furnishings produced past barbiturates are usually ascribed to their interference of nervus transmission to the cortex. Various theories for the activeness of barbiturates include: changes in ion movements across the jail cell membrane; interactions with cholinergic and non cholinergic receptor sites; impairment of biochemical reactions which provide energy; and depression of selected areas of the encephalon. The structures of the barbiturates can be related to the duration of effective activity. Although over 2000 derivatives of barbituric acid have been synthesized merely about a dozen are currently used. All of the barbiturates are related to the structure of barbituric acid shown beneath.
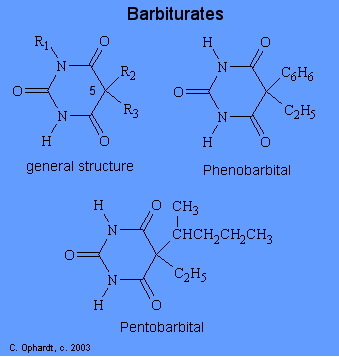
The duration of effect depends mainly on the alkyl groups attached to carbon # v which confer lipid solubility to the drug. The duration of effective activeness decreases every bit the total number of carbons at C # 5 increases. To be more specific, a long outcome is accomplished by a short chain and/or phenyl group. A curt duration effect occurs when at that place are the most carbons and branches in the alkyl chains
Benzodiazepines
The term benzodiazepine refers to the portion of the structure composed of a benzene ring (A) fused to a seven-membered diazepine ring (B). Nonetheless, since all of the important benzodiazepines contain a aryl substituent ring C) and a i, 4-diazepine ring, the term has come to mean the aryl-i,4-benzodiazepines. At that place are several useful benzodiazepines bachelor: chlordiazepoxide (Librium) and diazapam (Valium).
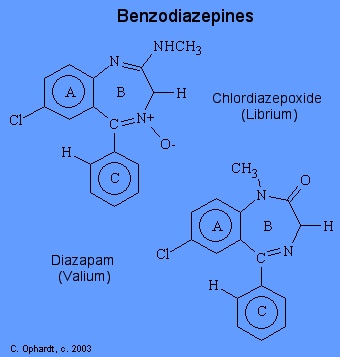
The deportment of benzodiazepines are a result of increased activation of receptors by gamma-aminobutyric acid (GABA). Benzodiazepine receptors are located on the alpha subunit of the GABA receptor located near exclusively on postsynaptic nerve endings in the CNS (especially cerebral cortex). Benzodiazepines enhance the GABA transmitter in the opening of postsynaptic chloride channels which leads to hyperpolarization of jail cell membranes. That is, they "bend" the receptor slightly so that GABA molecules adhere to and actuate their receptors more effectively and more oftentimes.
Contributors
Charles Ophardt (Professor Emeritus, Elmhurst Higher); Virtual Chembook
Source: https://chem.libretexts.org/Bookshelves/Biological_Chemistry/Supplemental_Modules_(Biological_Chemistry)/Pharmaceuticals/Barbiturates_and_Benzodiazepines
Posted by: jonespleamak1958.blogspot.com

0 Response to "What Is The Chemical Makeup Of Barbiturates"
Post a Comment